![PDF] Identification of potential surrogate end points in randomized clinical trials of aggressive and indolent non-Hodgkin's lymphoma: correlation of complete response, time-to-event and overall survival end points | Semantic Scholar PDF] Identification of potential surrogate end points in randomized clinical trials of aggressive and indolent non-Hodgkin's lymphoma: correlation of complete response, time-to-event and overall survival end points | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/1b7c7d2ac6a943299f03859ee0550a5a659900c1/4-Table2-1.png)
PDF] Identification of potential surrogate end points in randomized clinical trials of aggressive and indolent non-Hodgkin's lymphoma: correlation of complete response, time-to-event and overall survival end points | Semantic Scholar

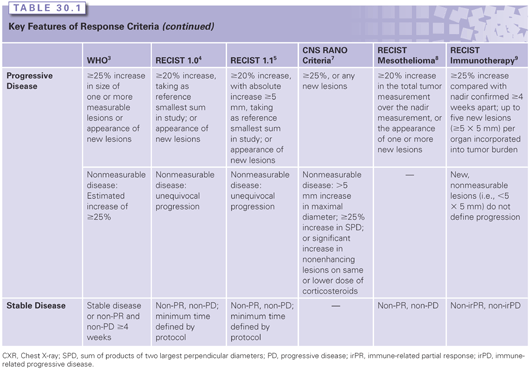
Supplemental Materials for iRECIST: guidelines for response criteria for use in trials testing immunotherapeutics - The Lancet Oncology

A randomized, multi-national, non-inferiority, phase III trial to evaluate the safety and efficacy of BF-200 ALA gel versus MAL cream in the treatment. - ppt download

Rituximab before splenectomy in adults with primary idiopathic thrombocytopenic purpura: a meta‐analysis - Auger - 2012 - British Journal of Haematology - Wiley Online Library

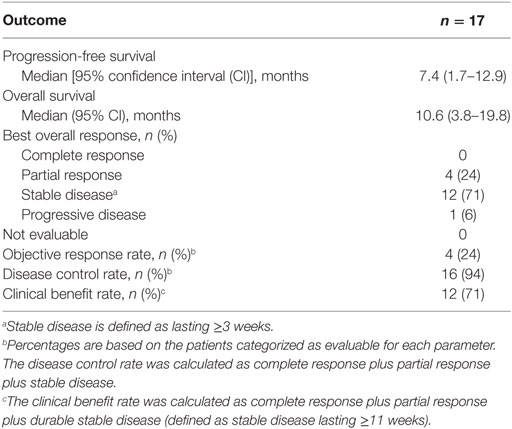
Investigator-assessed confirmed objective response rate (per RECIST),... | Download Scientific Diagram
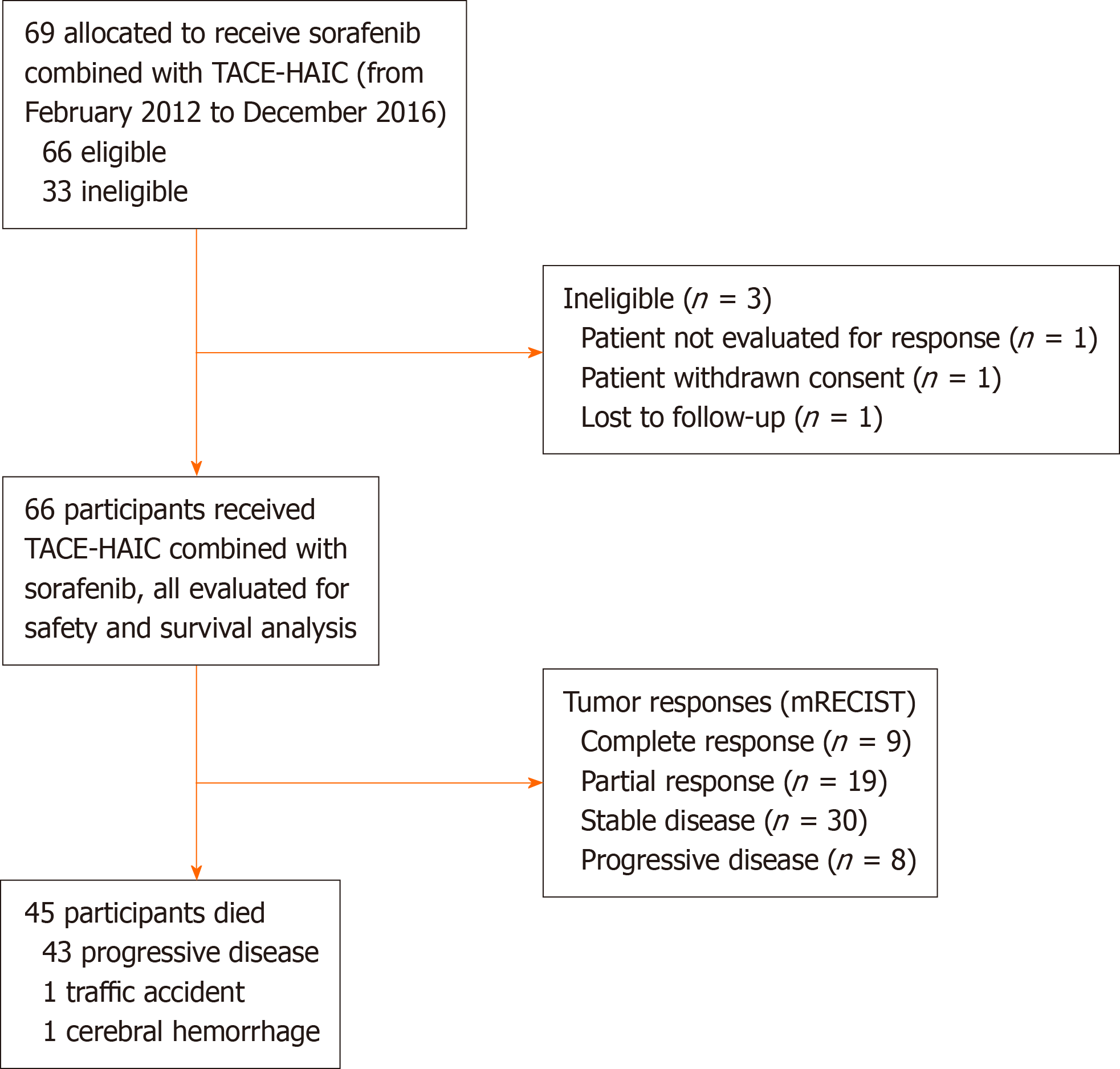
Sorafenib combined with embolization plus hepatic arterial infusion chemotherapy for inoperable hepatocellular carcinoma
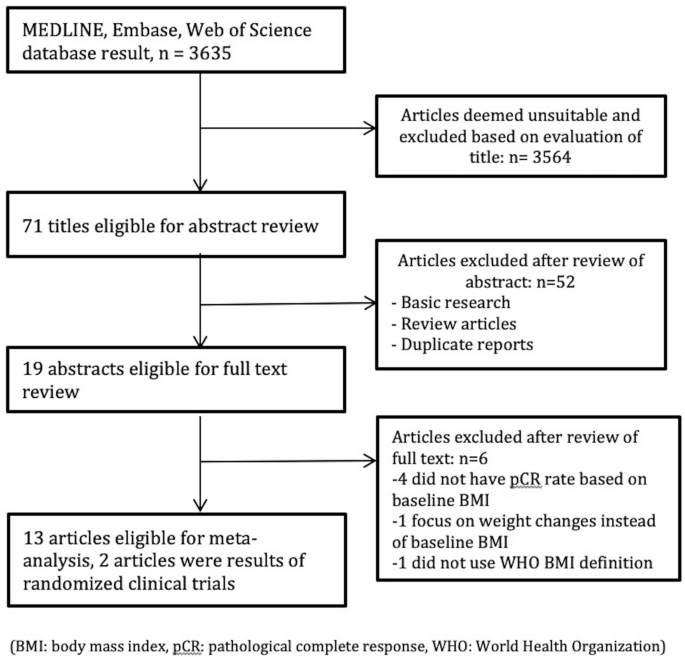
Impact of body mass index on pathological complete response following neoadjuvant chemotherapy in operable breast cancer: a meta-analysis | SpringerLink






![PDF] Evaluation and monitoring of response to therapy in multiple myeloma. | Semantic Scholar PDF] Evaluation and monitoring of response to therapy in multiple myeloma. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/c7433976b18e415f228f8d8e6521bba0c26bbc89/2-Table1-1.png)